Latest Science Note
Dojindo Science Note [Jun. 4, 2024]
|
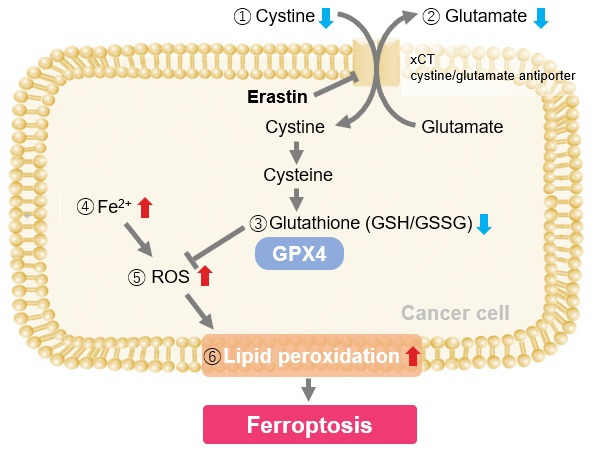
Ferroptosis, a form of regulated cell death driven by iron-dependent lipid peroxidation, is tightly controlled by several proteins. Key regulators include GPX4, which reduces lipid hydroperoxides with glutathione (GSH) and inhibits ferroptosis, and FSP1, which provides an alternative antioxidant defense by regenerating CoQ10. Other proteins, such as iron regulatory proteins and those involved in lipid metabolism, also modulate ferroptosis by influencing iron homeostasis and lipid peroxidation pathways. Disruption or dysregulation of these protein networks can either increase susceptibility to ferroptosis or confer resistance, highlighting their critical role in maintaining cellular health. |
||
|
Targeting SIRT3 sensitizes glioblastoma to ferroptosis by promoting mitophagy and inhibiting SLC7A11 |
Exosomal HSPB1, interacting with FUS protein, suppresses hypoxia-induced ferroptosis in pancreatic cancer by stabilizing Nrf2 mRNA and repressing P450 |
Regulation of FSP1 myristoylation by NADPH: A novel mechanism for ferroptosis inhibition |
|
Point of Interest - Inhibition of SIRT3 sensitizes GBM cells to RSL3-induced ferroptosis, leading to accumulation of iron and ROS in the mitochondria, which triggers mitophagy. -Inhibition of SIRT3 also downregulates transcription of SLC7A11, which increases cellular import of cystine. - Enforced expression of SLC7A11 in GBM cells with SIRT3 knockdown restores cellular cystine uptake and leads to cellular GSH levels. |
Point of Interest - In hypoxic cells incubated with the HSPB1-containing exosomes, cell proliferation and invasion are promoted and ferroptosis is suppressed. - Increased HSPB1 in cells leads to increased FUS expression and subsequently FUS stabilizes the mRNA of Nrf2, a known anti-ferroptosis gene. - The exosomes administration promotes tumor growth in nude mice, which can be suppressed by knockdown of HSPB1. |
Point of Interest - NMT2 modifies ferroptosis suppressor protein 1 (FSP1), a known free radical scavenging antioxidant. - NADPH increases N-myristoylated FSP1, which localizes to the plasma membrane and provides neurons with resistance to ferroptosis. |
| Related Techniques | ||
| Ferrous ion (Fe2+) detection | FerroOrange(intracellular), Mito-FerroGreen(mitochondrial) | |
| Lipid peroxidation detection | Liperfluo(intracellular), MitoPeDPP(mitochondrial) | |
| Total ROS detection | Highly sensitive DCFH-DA or Photo-oxidation Resistant DCFH-DA | |
| Mitochondrial superoxide detection | MitoBright ROS Deep Red - Mitochondrial Superoxide Detection | |
| Mitophagy or autophagy detection | Mitophagy Detection Kit, Autophagic Flux Assay Kit | |
| Glutathione Quantification | GSSG/GSH Quantification Kit | |
| Cystine Uptake detection | Cystine Uptake Assay Kit | |
| NAD(H) and NADP(H) redox couples assay | NAD/NADH and NADP/NADPH Assay Kit | |
| Cell Proliferation / Cytotoxicity Assay | Cell Counting Kit-8, Cytotoxicity LDH Assay Kit-WST | |
| Glycolysis/Oxidative phosphorylation Assay | Glycolysis/OXPHOS Assay Kit | |
| Related Applications | ||
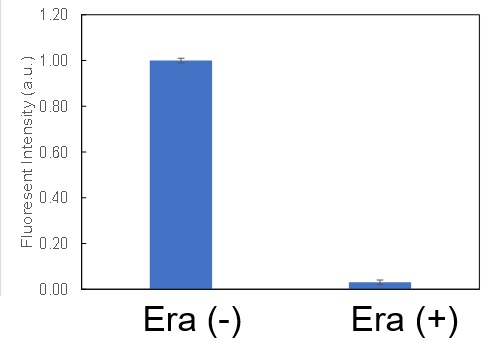
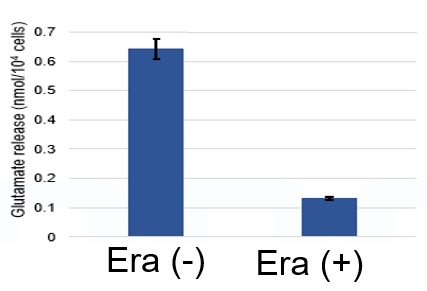
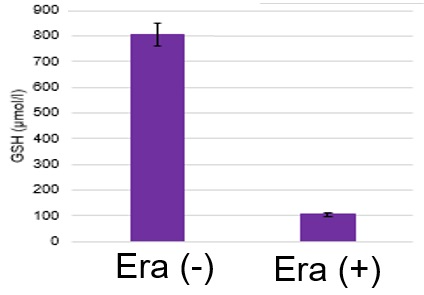
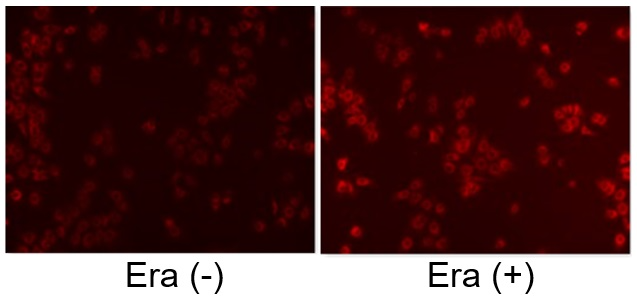
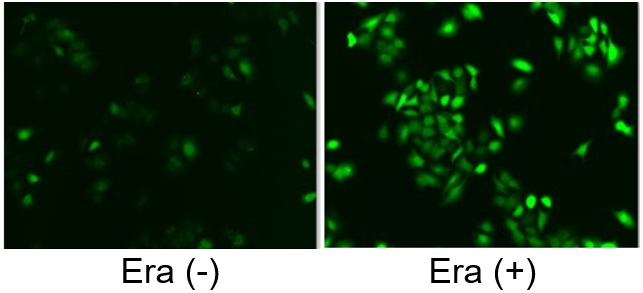
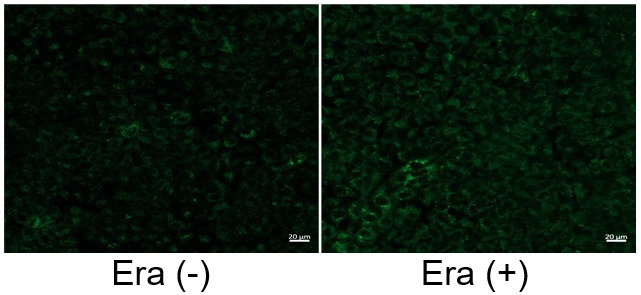
Induction of Ferroptosis by ErastinErastin is a known inducer of ferroptosis. By inhibiting the cystine transporter (xCT), erastin inhibits the uptake of cystine. Cystine is the raw material for GSH. Therefore, Erastin ultimately decreases the amount of GSH. Decreased GSH then results in lipid peroxide accumulation and induction of ferroptosis. Using erastin-treated A549 cells, we measured intracellular Fe2+, ROS, lipid peroxide, glutathione, glutamate release into the extracellular space, and cystine uptake. As a result, inhibition of xCT by elastin was observed and also the release of glutamate and uptake of cystine were decreased. Furthermore, elastin treatment decreased intracellular glutathione while it increased intracellular Fe2+ , ROS, and lipid peroxides.
|
||