TMBZ・HCl

Coloring Substrate for Peroxidase
-
Product codeT039 TMBZ・HCl
-
CAS No.207738-08-7
-
Chemical name3,3',5,5'-Tetramethylbenzidine, dihydrOchloride, dihydrate
-
MWC16H22Cl2N2・2H2O=349.30
| Unit size | Price | Item Code |
|---|---|---|
| 1 g | $191.00 | T039-10 |
Product Description
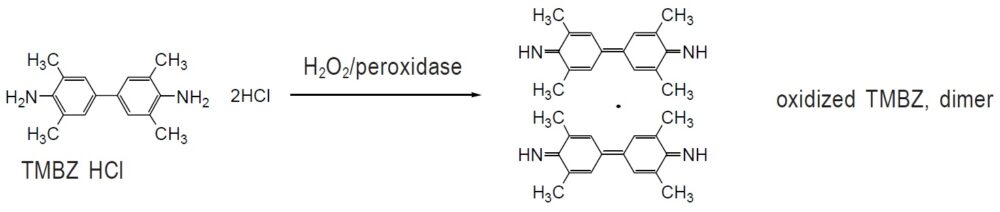
TMBZ is a chromogenic reagent utilized for peroxidase detection. It has been developed as an alternative to benzidine, which is a carcinogenic chemical. Because of the ortho methyl groups on its benzene ring, TMBZ is not metabolized into highly carcinogenic o-hydroxybenzidines or o,o E dihydroxybenzidines. Therefore, TMBZ compounds are much less carcinogenic than benzidine. Although the TMBZ solution is colorless, it turns bluish-green (λmax: 655 nm) in the presence of hydrogen peroxide and peroxidase. The structure of this bluish-green complex is thought to be a radical form of two oxidized TMBZ molecules. TMBZ HCl is a hydrochloride form of TMBZ that is readily soluble in water (100 mg TMBZ HCl per 10 ml water).
Technical info
1. Dissolve 6 mg of TMBZ with 1 ml DMSO to prepare 100X TMBZ solution.
2. Mix 5 μl of 30% hydrogen peroxide solution with 1 ml PBS to prepare 200X H2O2 solution.
3. Add 10 μl of 100X TMBZ solution and 5 μl of 200X H2O2 solution to 1 ml PBS to prepare staining solution.*
4. Add 100-200 μl of the staining solution to each well of a 96-well microplate. Incubate the plate at room temperature or at 37ºC for 5 minutes to 1 hour.
5. Wash the sample with PBS several times to stop the staining reaction.
* For the best results, modification of the final concentration of TMBZ and hydrogen peroxide may be necessary. The staining solution is not stable. Please prepare fresh solution prior to use.
References
1. V. R. Holland, et al., A Safer Substitute for Benzidine in the Detection of Blood. Tetrahedron. 1974;30:3299-3302.
2. W. Levin, et al., Preparation of Partially Purified, Lipid-depleted Cytochrome P-450 and Reduced Nicotinamide Adenine Dinucleotide Phosphatecytochrome c Reductase from Rat Liver Microsomes. J Biol Chem. 1974;249:1747-1754.
3. P. E. Thomas, et al., An Improved Staining Procedure for the Detection of the Peroxidase Activity of Cytochrome p-450 on Sodium Dodecyl Sulfate Polyacrylamide Gels. Anal Biochem. 1976;75:168-176.
4. H. H. Liem, et al., Quantitative Determination of Hemoglobin and Cytochemical Staining for Peroxidase Using 3, 3 E 5, 5 ETetramethylbenzidine dihydrochloride, a Safe Substitute for Benzidine. Anal Biochem. 1979;98:388-393.
5. R. C. Lijana, et al., Tetramethylbenzidine- A substitute for Benzidine in Hemoglobin Analysis. J Lab Clin Med. 1979;94:266-276.
6. R. M. Jaffe, et al., A New Occult Blood Test Not Subject to False-Negative Results from Reducing Substances. J Lab Clin Med. 1979;93:879-886.
7. K. Suzuki, et al., Assay Method for Myeloperoxidase in Human Polymorphonuclear Leukocytes. Anal Biochem. 1983;132:345-352.
8. F. H. Pujol, et al., A Double Sandwich Monoclonal Enzyme Immunoassay for Detection of Hepatitis B Surface Antigen. J Immunoassay. 1993;14:21-31.
9. F. B. Serrat, Colorimetric Method for Determiantion of Chlorine with 3, 3 E 5, 5 ETetramethylbenzidine. Talanta. 1994;41:2091-2094.
Handling and storage condition
| Appearance: | White to slightly pink crystalline powder |
|---|---|
| Purity (Titration): | ≧ 98.0 % |
| Solubility in water: | To pass test (clear, colorless to almost colorless) |
| pH(25°C): | 2.0 - 2.5 (25oC) |
| Water content: | 8.0 - 12.0 % |
| IR spectrum: | Authentic |
| Protect from light |













